While continuous positive airway pressure (CPAP) therapy is a proven method of treating obstructive sleep apnea (OSA), therapy noncompliance remains a major issue. Though still in the clinical trial stage in the U.S., continuous negative external pressure (cNEP) devices offer a potential alternative to CPAP for people with OSA.
We’ll take a look at what a cNEP device is and how it’s designed to alleviate OSA symptoms. We’ll also explore how it differs from CPAP therapy and answer some frequently asked questions about cNEP technology.
What Are cNEP Devices?
cNEP devices are a noninvasive therapy for treating OSA. They attach to the outside of the neck and apply continuous negative pressure. This suction effect is designed to pull the front of the neck away from the back of the throat, opening the airway.
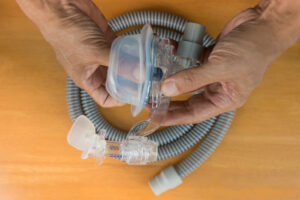
cNEPs differ from CPAP machines, which deliver pressurized air to the user’s airway through a hose that connects to a face mask. cNEPs are designed to offer a maskless alternative for people with OSA who have trouble tolerating positive pressure therapy.
How cNEP Devices Alleviate OSA Symptoms
Obstructive sleep apnea is a condition characterized by pauses in breathing during sleep due to a blocked or narrowed airway. It occurs when the muscles in the throat relax, leading to upper airway collapse.
cNEP devices contain an air pump attached to a silicone collar that forms a seal against the front of the neck. The pump and collar work together to create exterior negative pressure, a gentle suctioning effect that’s engineered to pull the front of the neck forward to prevent the soft tissues in the throat from collapsing.
What Separates cNEP From PAP Therapy?
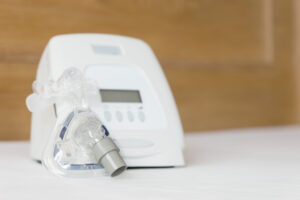
Traditional CPAP machines contain an air compressor, an air filter, a control panel, and a power cord. A flexible tube attached to the device delivers pressurized air to a CPAP mask.
cNEP devices are battery operated and rechargeable. They have a silicone collar that fits snugly against the front of the neck and the bottom of the chin. A small pump on the front of the device acts as a vacuum to create negative external pressure.
The lack of a cord, tube, mask, and bedside machine make cNEPs the more compact option, with easier maintenance and cleaning. Since there’s no mask component, cNEP devices could also eliminate common CPAP side effects like dry mouth and nasal discomfort.
Is cNEP an Effective Treatment for OSA Symptoms?
While CPAP has been used as a sleep apnea treatment for several decades, cNEP devices have yet to be approved in the U.S. and few clinical studies have been completed.
In one pilot study, researchers found that 60% of participants showed excellent response to cNEP therapy, while another 27% had a partial response. However, the study involved just 15 people. Two study participants developed a mild rash where the device contacted the skin, and one person had mild blistering.
Another slightly larger study revealed mixed results. While 64% of participants displayed a sustained response to cNEP, three people saw an increase in OSA severity during an in-lab sleep study. Similar side effects were reported.
Frequently Asked Questions About cNEP
cNEP is not currently approved by the Food and Drug Administration as an OSA treatment. However, a cNEP device has received a Breakthrough Device designation from the FDA, which could potentially fast-track its approval process. That device is in the clinical trial stage, with an estimated study completion in September 2023.
cNEP devices are not currently approved for purchase in the United States. Since any device used to treat sleep apnea is considered a medical device, a cNEP product would first need FDA approval before it could be sold to people with a doctor’s prescription.
Iron lungs and cNEP devices use negative pressure to aid breathing, but the similarities end there. An iron lung is a cylindrical chamber that encloses a person’s entire body except their head. The air pressure inside the respirator is alternately decreased and increased to manually cause the lungs to inflate and deflate when a person has difficulty breathing on their own. They were often used during the early 20th century for people with polio and others requiring breathing assistance.
Get Your Sleep Questions Answered Live on 4/30
Have questions about sleep? Get all your sleep-related questions answered in a Live Q&A on YouTube with renowned sleep expert Dr. Michael Breus at 5 p.m. PST/8 p.m. EST.